The Identification of Medicinal Products (IDMP) is a set of five ISO data standards requiring companies to provide and maintain specific data regarding their products in an electronic format and ensure it’s always up to date. It establishes an internationally harmonized way to represent medicinal products. The data required by IDMP will create unified safety standards, product descriptions and usage information, and assure common messaging standards for global understanding.
Why IDMP?
IDMP’s goal is to standardize drug product identification around the globe in order to increase safety reporting’s accuracy. To be IDMP compliant, biopharma organizations will be compelled to implement vast changes to their product processes. New solutions enhancing cross-functional collaboration will be required to assure compliance. IDMP’s scope is broad, requiring information about manufacturers, marketing authorization holders, packaging production details, substance and ingredient lists, and more. Due to IDMP’s enormity, most countries are expected to implement changes gradually.
What This Means for Pharma Companies
Pharma companies work with a mind-boggling amount of content. This content, data by any other name, must be represented per IDMP consistently. Within regulatory affairs, collecting and understanding the detailed registered particulars and establishing processes that collect the data points for proposed changes to accompany submissions must be created. Companies must therefore represent not only the current approved registered particulars, but also the proposed registered particulars.
While companies have various systems and methods for collecting and managing this data, they often work in disparate technology systems. As such, they may find themselves having the business processes but not the connected technology to support it. This is especially more likely for small and midsize companies. Such companies may find it advantageous to invest in solutions that will enhance their abilities to comply with IDMP and increase their likelihood of maintaining their positions as market leaders.
So, how are companies going to wrap their arms around the new regulation? Most of the data companies currently work with resides in unstructured documentation. IDMP will now require this data to be captured in authoritative structured systems.
Managing Product Information According to IDMP
It goes without saying data must always be accurate. To maintain correct information, data needs to be stored, updated, and managed in a way that everyone knows is up to date, consistent, and correct. This is where software providers come in, providing direction for companies that need a place to start.
To comply with regulatory requirements and assure the quality and safety of pharmaceutical goods, change management in the pharmaceutical sector, particularly for regulatory affairs teams, entails implementing and managing changes to procedures, systems, and documentation. This involves recognizing, evaluating, and communicating prospective changes, creating and implementing sensible procedures and controls, and making sure that any changes are properly documented and examined by regulatory authorities.
Teams in charge of regulatory affairs oversee the regulatory elements of pharmaceutical products at every stage of research and approval, as well as post-market monitoring. When it comes to change management, they must ensure any modifications made to a product or procedure adhere to all applicable regulatory standards, including those for quality, safety, and efficacy.
The pharmaceutical sector needs an organized and methodical approach to change management that includes cross-functional cooperation, stakeholder participation, effective communication, and best practices for documentation. It also calls for a thorough comprehension of regulatory standards and the capacity to negotiate intricate regulatory systems on a national and worldwide scale.
Automating Processes for More Efficiency
Pharmaceutical businesses may streamline their operations with the help of automation, which also ensures the accuracy and quality of their products. Companies can decrease errors and boost productivity by automating repetitive and normal processes like data input and document management. This frees up staff to work on more complex projects. By imposing consistent practices and controls, such as those related to product testing and validation, automation can also help businesses make sure their goods comply with regulatory standards.
Automation can also increase content accuracy by enabling team collaboration and communication, as well as real-time access to product information. Overall, automation can benefit pharmaceutical businesses by saving time, lowering costs, and enhancing the quality and accuracy of their products, both of which are crucial for upholding regulatory compliance and satisfying clients’ and patients’ expectations.
A Role for Structured Content Management
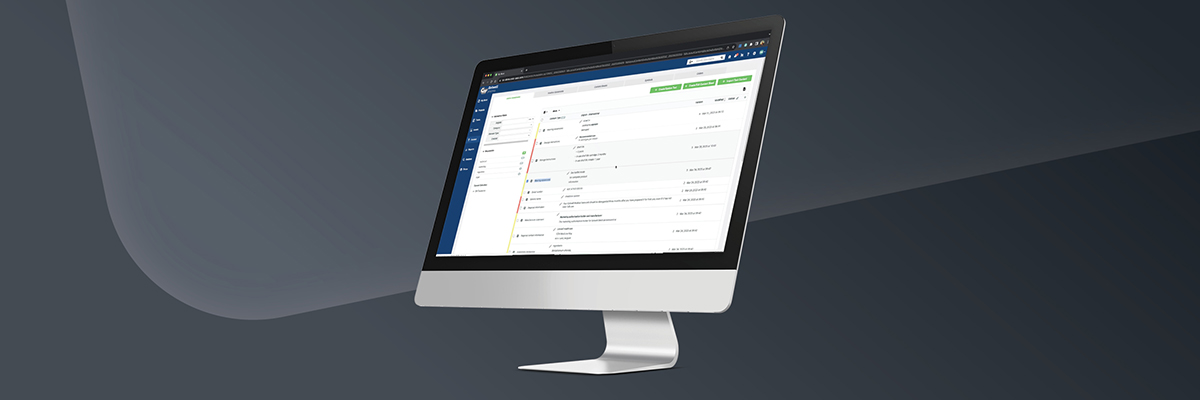
Structured Content Management (SCM) solutions play an important role in the implementation of Identification of Medicinal Products (IDMP) requirements. Structured Content Management, a module in Esko’s software platform for Life Sciences, helps ensure the data elements required for IDMP compliance are managed and maintained in a consistent, accurate, and efficient manner. Here are some of the ways in which SCM solutions help with IDMP implementation:
- Automated data standardization: Automatically standardize the naming conventions, coding systems, and terminologies used for medicinal products, ingredients, routes of administration, and other key elements required for IDMP compliance. This ensures consistency and accuracy in the data and reduces the risk of errors or confusion.
- Automated data integration: Integrate data from various sources, such as clinical trial data, regulatory submissions, and manufacturing information, to create a comprehensive and accurate view of a medicinal product’s data. This facilitates the submission of complete and consistent IDMP data sets.
- Centralized data management: With SCM, companies can manage and maintain the data required for IDMP compliance in a central repository, enabling companies to store, access, and update data efficiently.
- Automated data mapping: Automated mapping of data elements between different regulatory authorities ensures data is consistent and compliant across all jurisdictions.
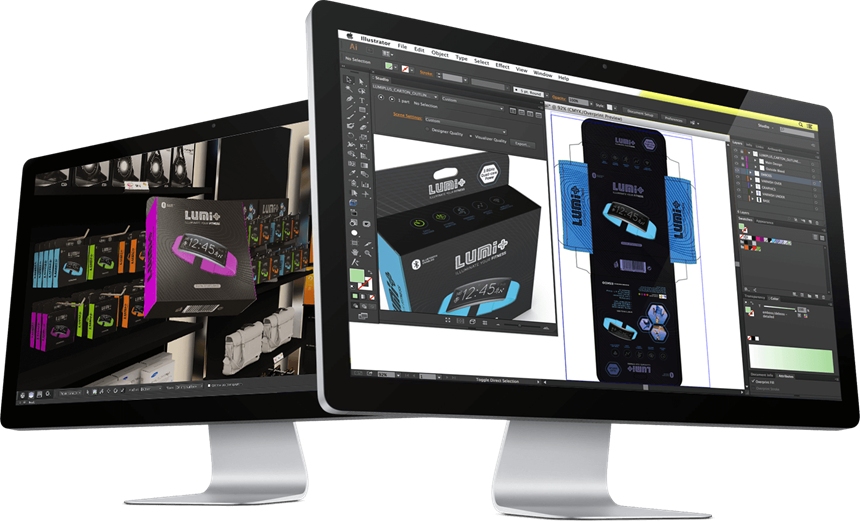
- Automated output generation: Generate output in IDMP standard automatically, eliminating the need for manual data entry and reducing the risk of errors or omissions. Additionally, SCM produces output for printed materials such as labels, packaging, and Inserts based on templates, ensuring consistency in branding and content.
Structured content management can automatically produce output in IDMP standards, ensuring compliance and reducing risk. Overall, SCM represents the future of best practices for pharmaceutical professionals.
IDMP is the Future
In sum, ISO’s IDMP is a global regulatory framework for the standardization, identification, documentation, and exchange of pharmaceutical product information. The regulation is designed to improve patient safety, maximize pharmacovigilance, and ensure regulatory compliance across international borders. It requires companies to submit detailed information about their products, including unique identification codes, manufacturing information, ingredients, and dosage forms, to regulatory authorities. This information is then stored in a centralized database, enabling better collaboration, and sharing of data among regulatory agencies. The regulation is set to be implemented gradually over the next few years.
IDMP is here to stay, and pharmaceutical companies need to allocate resources to achieve compliance with this new regulatory framework. Contact Esko to learn more about structured content management and how it will help your company.